Vitamin C ((L-Ascorbic Acid DC) granulation)

City: LV, Marupe
Please avoid sudden changes in temperature and humidity, as they may affect the condition of derivative ingredients and tablets.
The shelf life begins from the date of manufacture at the factory.
Taking into account logistical and transportation stages, the remaining shelf life of the product is always shorter than the full shelf life.
We ship products to our customers with a remaining shelf life of no less than 70%.
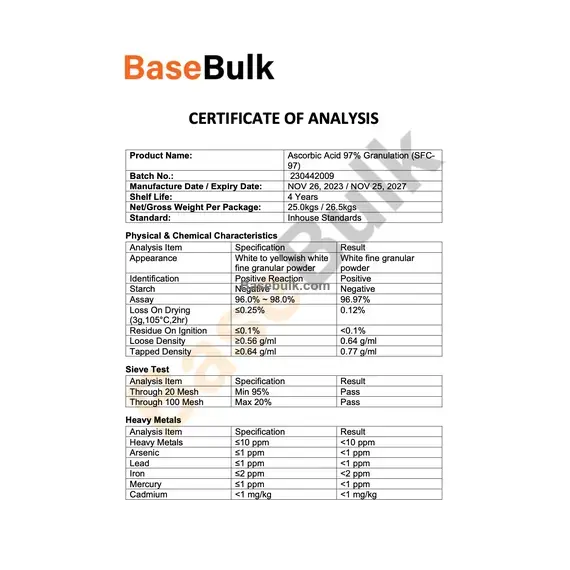
Physical & Chemical Characteristics
Analysis Item | Specification | Result |
Appearance | White to yellowish white fine granular powder | White fine granular powder |
Identification | Positive Reaction | Positive |
Starch | Negative | Negative |
Assay | 96.0% ~ 98.0% | 96.97% |
Loss On Drying (3g,105°C,2hr) | ≤0.25% | 0.12% |
Residue On Ignition | ≤0.1% | <0.1% |
Loose Density | ≥0.56 g/ml | 0.64 g/ml |
Tapped Density | ≥0.64 g/ml | 0.77 g/ml |
Sieve Test
Analysis Item | Specification | Result |
Through 20 Mesh | Min 95% | Pass |
Through 100 Mesh | Max 20% | Pass |
Heavy Metals
Analysis Item | Specification | Result |
Heavy Metals | ≤10 ppm | <10 ppm |
Arsenic | ≤1 ppm | <1 ppm |
Lead | ≤1 ppm | <1 ppm |
Iron | ≤2 ppm | <2 ppm |
Mercury | ≤1 ppm | <1 ppm |
Cadmium | <1 mg/kg | <1 mg/kg |
Microbiological Tests
Analysis Item | Specification | Result |
Total Aerobic Microbial Count | ≤500 CFU/g | <500 CFU/g |
Moulds and Yeast Count | ≤100 CFU/g | <100 CFU/g |