Microcrystalline Cellulose PH-200

City: LV, Marupe
Please avoid sudden changes in temperature and humidity, as they may affect the condition of derivative ingredients and tablets.
The shelf life begins from the date of manufacture at the factory.
Taking into account logistical and transportation stages, the remaining shelf life of the product is always shorter than the full shelf life.
We ship products to our customers with a remaining shelf life of no less than 70%.
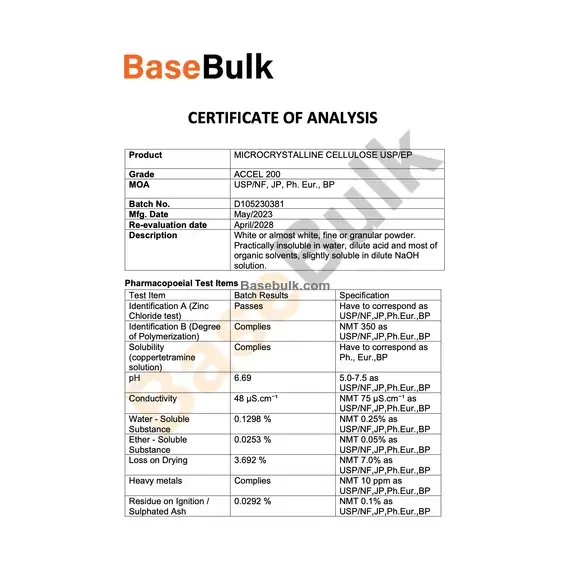
Pharmacopoeial Test Items
Test Item | Batch Results | Specification |
Identification A (Zinc Chloride test) | Passes | Have to correspond as USP/NF,JP,Ph.Eur.,BP |
Identification B (Degree of Polymerization) | Complies | NMT 350 as USP/NF,JP,Ph.Eur.,BP |
Solubility (coppertetramine solution) | Complies | Have to correspond as Ph., Eur.,BP |
pH | 6.69 | 5.0-7.5 as USP/NF,JP,Ph.Eur.,BP |
Conductivity | 48 µS.cm⁻¹ | NMT 75 µS.cm⁻¹ as USP/NF,JP,Ph.Eur.,BP |
Water - Soluble Substance | 0.1298% | NMT 0.25% as USP/NF,JP,Ph.Eur.,BP |
Ether - Soluble Substance | 0.0253% | NMT 0.05% as USP/NF,JP,Ph.Eur.,BP |
Loss on Drying | 3.692% | NMT 7.0% as USP/NF,JP,Ph.Eur.,BP |
Heavy metals | Complies | NMT 10 ppm as USP/NF,JP,Ph.Eur.,BP |
Residue on Ignition / Sulphated Ash | 0.0292% | NMT 0.1% as USP/NF,JP,Ph.Eur.,BP |
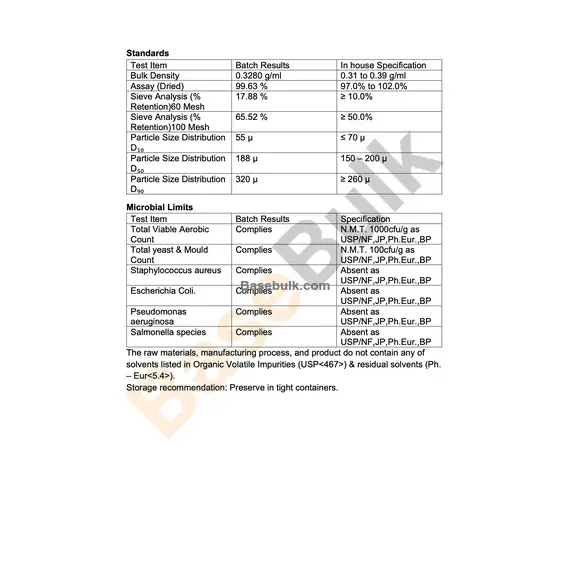
Standards
Test Item | Batch Results | In house Specification |
Bulk Density | 0.3280 g/ml | 0.31 to 0.39 g/ml |
Assay (Dried) | 99.63% | 97.0% to 102.0% |
Sieve Analysis (% Retention)60 Mesh | 17.88 % | ≥ 10.0% |
Sieve Analysis (% Retention)100 Mesh | 65.52% | ≥ 50.0% |
Particle Size Distribution D₁₀ | 55 µ | ≤ 70 µ |
Particle Size Distribution D₅₀ | 188 µ | 150 – 200 µ |
Particle Size Distribution D₉₀ | 320 µ | ≥ 260 µ |
Microbial Limits
Test Item | Batch Results | Specification |
Total Viable Aerobic Count | Complies | N.M.T. 1000cfu/g as USP/NF,JP,Ph.Eur.,BP |
Total yeast & Mould Count | Complies | N.M.T. 100cfu/g as USP/NF,JP,Ph.Eur.,BP |
Staphylococcus aureus | Complies | Absent as USP/NF,JP,Ph.Eur.,BP |
Escherichia Coli. | Complies | Absent as USP/NF,JP,Ph.Eur.,BP |
Pseudomonas aeruginosa | Complies | Absent as USP/NF,JP,Ph.Eur.,BP |
Salmonella species | Complies | Absent as USP/NF,JP,Ph.Eur.,BP |
The raw materials, manufacturing process, and product do not contain any of solvents listed in Organic Volatile Impurities (USP<467>) & residual solvents (Ph. – Eur<5.4>). Storage recommendation: Preserve in tight containers.