Hawthorn Berry FRUIT

City: LV, Marupe
Please avoid sudden changes in temperature and humidity, as they may affect the condition of derivative ingredients and tablets.
The shelf life begins from the date of manufacture at the factory.
Taking into account logistical and transportation stages, the remaining shelf life of the product is always shorter than the full shelf life.
We ship products to our customers with a remaining shelf life of no less than 70%.
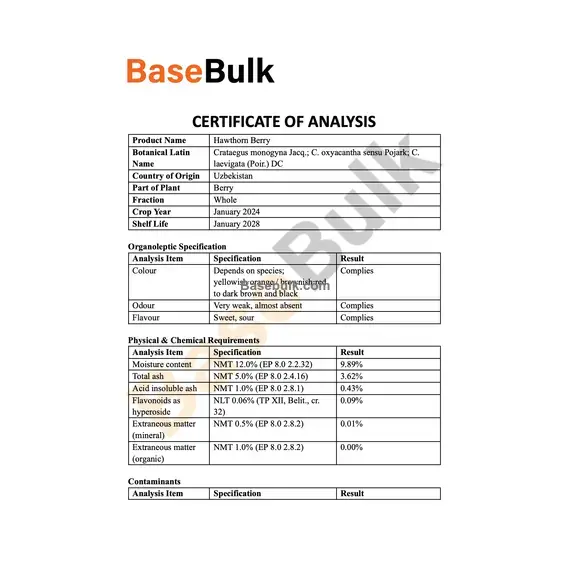
Organoleptic Specification
Analysis Item | Specification | Result |
Colour | Depends on species; yellowish‑orange / brownish‑red to dark brown and black | Complies |
Odour | Very weak, almost absent | Complies |
Flavour | Sweet, sour | Complies |
Physical & Chemical Requirements
Analysis Item | Specification | Result |
Moisture content | NMT 12.0% (EP 8.0 2.2.32) | 9.89% |
Total ash | NMT 5.0% (EP 8.0 2.4.16) | 3.62% |
Acid insoluble ash | NMT 1.0% (EP 8.0 2.8.1) | 0.43% |
Flavonoids as hyperoside | NLT 0.06% (TP XII, Belit., cr. 32) | 0.09% |
Extraneous matter (mineral) | NMT 0.5% (EP 8.0 2.8.2) | 0.01% |
Extraneous matter (organic) | NMT 1.0% (EP 8.0 2.8.2) | 0.00% |
Contaminants
Analysis Item | Specification | Result |
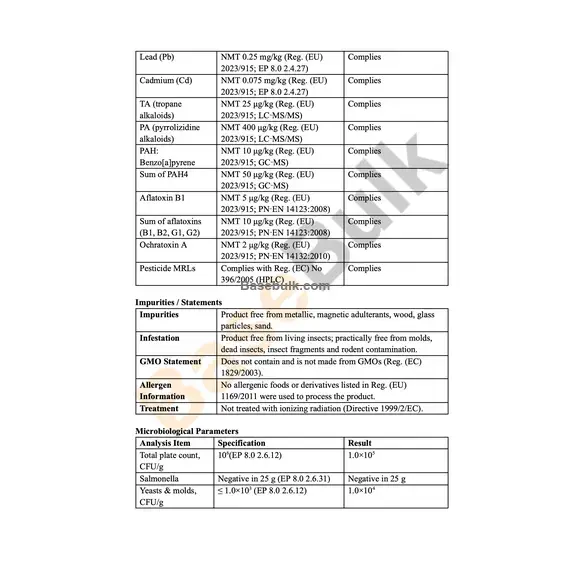
Lead (Pb) | NMT 0.25 mg/kg (Reg. (EU) 2023/915; EP 8.0 2.4.27) | Complies |
Cadmium (Cd) | NMT 0.075 mg/kg (Reg. (EU) 2023/915; EP 8.0 2.4.27) | Complies |
TA (tropane alkaloids) | NMT 25 μg/kg (Reg. (EU) 2023/915; LC‑MS/MS) | Complies |
PA (pyrrolizidine alkaloids) | NMT 400 μg/kg (Reg. (EU) 2023/915; LC‑MS/MS) | Complies |
PAH: Benzo[a]pyrene | NMT 10 μg/kg (Reg. (EU) 2023/915; GC‑MS) | Complies |
Sum of PAH4 | NMT 50 μg/kg (Reg. (EU) 2023/915; GC‑MS) | Complies |
Aflatoxin B1 | NMT 5 μg/kg (Reg. (EU) 2023/915; PN‑EN 14123:2008) | Complies |
Sum of aflatoxins (B1, B2, G1, G2) | NMT 10 μg/kg (Reg. (EU) 2023/915; PN‑EN 14123:2008) | Complies |
Ochratoxin A | NMT 2 μg/kg (Reg. (EU) 2023/915; PN‑EN 14132:2010) | Complies |
Pesticide MRLs | Complies with Reg. (EC) No 396/2005 (HPLC) | Complies |
Impurities / Statements
Impurities | Product free from metallic, magnetic adulterants, wood, glass particles, sand. |
Infestation | Product free from living insects; practically free from molds, dead insects, insect fragments and rodent contamination. |
GMO Statement | Does not contain and is not made from GMOs (Reg. (EC) 1829/2003). |
Allergen Information | No allergenic foods or derivatives listed in Reg. (EU) 1169/2011 were used to process the product. |
Treatment | Not treated with ionizing radiation (Directive 1999/2/EC). |
Microbiological Parameters
Analysis Item | Specification | Result |
Total plate count, CFU/g | 106(EP 8.0 2.6.12) | 1.0×105 |
Salmonella | Negative in 25 g (EP 8.0 2.6.31) | Negative in 25 g |
Yeasts & molds, CFU/g | ≤ 1.0×103 (EP 8.0 2.6.12) | 1.0×104 |
E. coli / g | ≤ 10 (EP 8.0 2.6.31) | < 10 |
Packaging & Storage
Shelf Life | 48 months (in original packing and proper storage). |
Labeling | Product name, origin, net weight, gross weight, expiry date, batch no. |