L-Carnitine

City: LV, Marupe
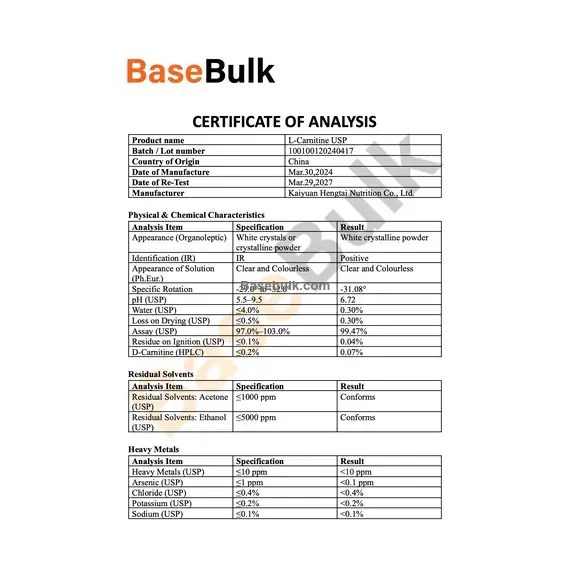
Physical & Chemical Characteristics
Analysis Item | Specification | Result |
Appearance (Organoleptic) | White crystals or crystalline powder | White crystalline powder |
Identification (IR) | IR | Positive |
Appearance of Solution (Ph.Eur.) | Clear and Colourless | Clear and Colourless |
Specific Rotation | -29.0° to -32.0° | -31.08° |
pH (USP) | 5.5–9.5 | 6.72 |
Water (USP) | ≤4.0% | 0.30% |
Loss on Drying (USP) | ≤0.5% | 0.30% |
Assay (USP) | 97.0%–103.0% | 99.47% |
Residue on Ignition (USP) | ≤0.1% | 0.04% |
D-Carnitine (HPLC) | ≤0.2% | 0.07% |
Residual Solvents
Analysis Item | Specification | Result |
Residual Solvents: Acetone (USP) | ≤1000 ppm | Conforms |
Residual Solvents: Ethanol (USP) | ≤5000 ppm | Conforms |
Heavy Metals
Analysis Item | Specification | Result |
Heavy Metals (USP) | ≤10 ppm | <10 ppm |
Arsenic (USP) | ≤1 ppm | <0.1 ppm |
Chloride (USP) | ≤0.4% | <0.4% |
Potassium (USP) | ≤0.2% | <0.2% |
Sodium (USP) | ≤0.1% | <0.1% |
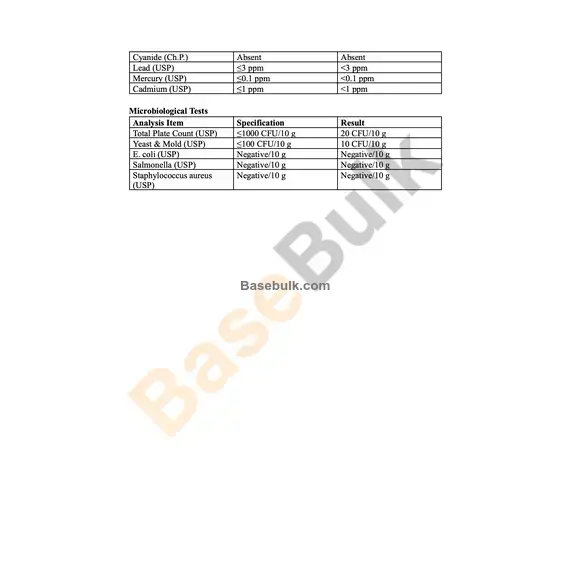
Cyanide (Ch.P.) | Absent | Absent |
Lead (USP) | ≤3 ppm | <3 ppm |
Mercury (USP) | ≤0.1 ppm | <0.1 ppm |
Cadmium (USP) | ≤1 ppm | <1 ppm |
Microbiological Tests
Analysis Item | Specification | Result |
Total Plate Count (USP) | ≤1000 CFU/10 g | 20 CFU/10 g |
Yeast & Mold (USP) | ≤100 CFU/10 g | 10 CFU/10 g |
E. coli (USP) | Negative/10 g | Negative/10 g |
Salmonella (USP) | Negative/10 g | Negative/10 g |
Staphylococcus aureus (USP) | Negative/10 g | Negative/10 g |