DigeZyme

City: LV, Marupe
Please avoid sudden changes in temperature and humidity, as they may affect the condition of derivative ingredients and tablets.
The shelf life begins from the date of manufacture at the factory.
Taking into account logistical and transportation stages, the remaining shelf life of the product is always shorter than the full shelf life.
We ship products to our customers with a remaining shelf life of no less than 70%.
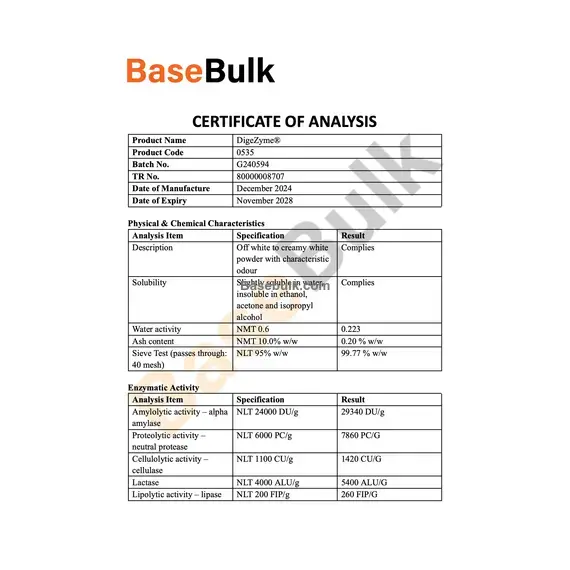
Physical & Chemical Characteristics
Analysis Item | Specification | Result |
Description | Off white to creamy white powder with characteristic odour | Complies |
Solubility | Slightly soluble in water, insoluble in ethanol, acetone and isopropyl alcohol | Complies |
Water activity | NMT 0.6 | 0.223 |
Ash content | NMT 10.0% w/w | 0.20 % w/w |
Sieve Test (passes through: 40 mesh) | NLT 95% w/w | 99.77 % w/w |
Enzymatic Activity
Analysis Item | Specification | Result |
Amylolytic activity – alpha amylase | NLT 24000 DU/g | 29340 DU/g |
Proteolytic activity – neutral protease | NLT 6000 PC/g | 7860 PC/G |
Cellulolytic activity – cellulase | NLT 1100 CU/g | 1420 CU/G |
Lactase | NLT 4000 ALU/g | 5400 ALU/G |
Lipolytic activity – lipase | NLT 200 FIP/g | 260 FIP/G |
Heavy Metals
Analysis Item | Specification | Result |
Lead | NMT 3 ppm (µg/g) | <0.20 ppm (µg/g) |
Arsenic | NMT 1 ppm (µg/g) | <0.20 ppm (µg/g) |
Cadmium | NMT 1 ppm (µg/g) | <0.20 ppm (µg/g) |
Mercury | NMT 0.1 ppm (µg/g) | <0.02 ppm (µg/g) |
Residual Solvents
Analysis Item | Specification | Result |
Residual Solvents: Ethylene oxide + 2-Chloroethanol | NMT 10 ppb (µg/kg) | Complies |
Contaminants
Analysis Item | Specification | Result |
Chloramphenicol (MRPL) by LC-MS/MS | NMT 0.3 ppb (µg/kg) | Complies |
Microbiological Tests
Analysis Item | Specification | Result |
Total aerobic microbial count | NMT 5000 cfu/g | 975 cfu/g |
Total yeasts and molds count | NMT 100 cfu/g | <10 cfu/g |
Escherichia coli | Negative/10 g | Complies |
Salmonella | Negative/10 g | Complies |
Staphylococcus aureus | Negative/10 g | Complies |
Pseudomonas aeruginosa | Negative/10 g | Complies |
Bile tolerant gram negative bacteria | NMT 100 cfu/g | <10 cfu/g |
Coliforms | Less than 10 cfu/g | <10 cfu/g |
General Status
Category | Intended for Nutraceutical application |
Preparation type | Multienzyme complex |
Standardization | Enzymatic activity |
Excipient used | Maltodextrin 70% to 78% (CAS 9050-36-6) |
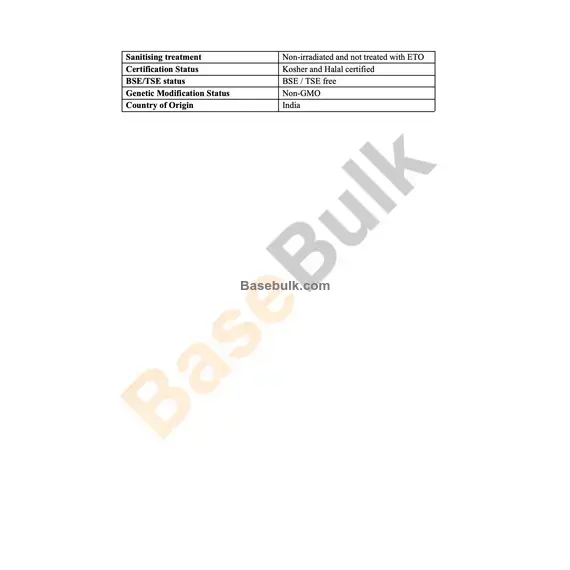
Sanitising treatment | Non-irradiated and not treated with ETO |
Certification Status | Kosher and Halal certified |
BSE/TSE status | BSE / TSE free |
Genetic Modification Status | Non-GMO |
Country of Origin | India |